Parkinson’s
Disease
Meet Hybridopa™
The world’s first Hybrid Drug™ for Parkinson’s Disease. Combining Levodopa with a clinically validated and mechanistically proven digital protocol to improve quality of life.
Clinical Milestone:
Phase 3 Ready
Following a successful Phase 2 trial where Hybridopa, the pioneering drug + Software as a Medical Device (SaMD) combination,
met its primary endpoint, we are launching our Phase 3 trial in H2 2026.
Approval
The "Levodopa Ceiling"
Over 10 million people worldwide are living with Parkinson’s Disease (PD), and nearly 90,000 people are newly diagnosed in the US with PD each year. It is a complicated neurodegenerative disease, and patients show a broad and diverse range of symptoms as the disease progresses.
Levodopa remains the gold standard for symptom management. As the disease progresses, patients face "wearing off" effects, freezing of gait, and speech deterioration that the drug alone cannot address.
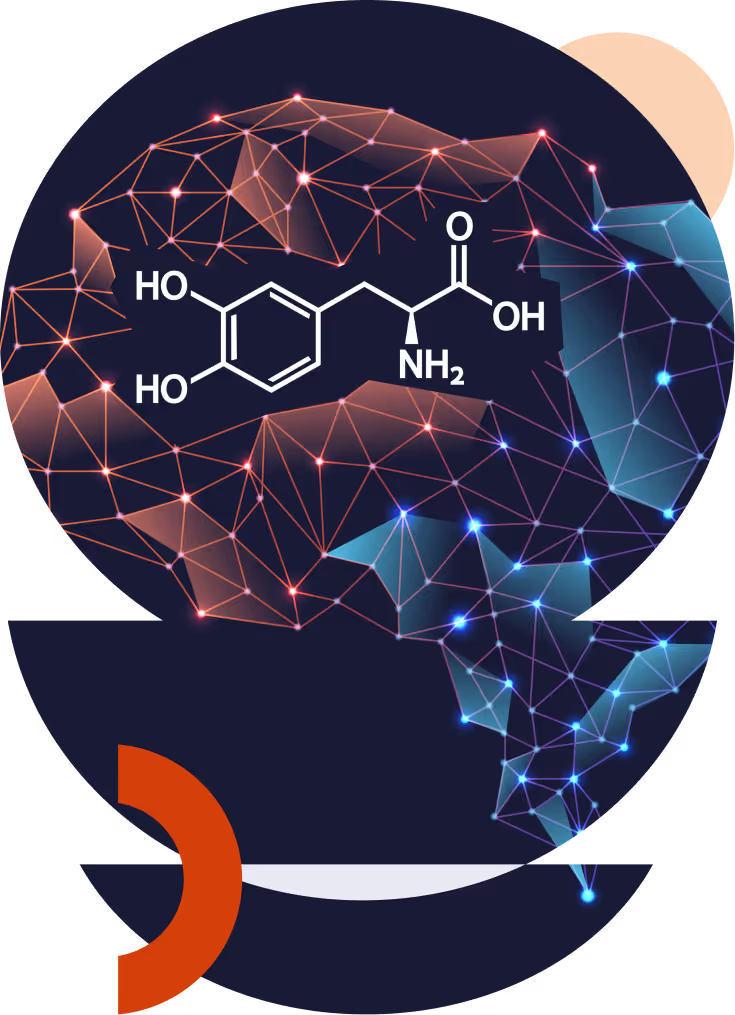
Active Neural Reinforcement via Digital Interactions
Hybridopa™ is designed to enhance the impact of Levodopa and help retrain the brain.
Hybridopa™ combines immediate-release levodopa/carbidopa with DopApp™, a prescription digital intervention delivered through a mobile application. The therapy uses adaptive protocols that respond to patient performance, behavior, and clinical status to enhance both motor and non-motor outcomes in Parkinson’s disease.
01 The Drug
Delivers necessary dopamine replacement.
02 The Digital Layer
A personalized, AI-driven protocol delivered via app to target the "Behavioral-Digital Pathway."
The Outcome
By synchronizing drug delivery with motor and cognitive training (gait, voice, executive function), we aim to maximize the functional benefit of every dose.
Primary Hybridopa Targets
Reduce Systemic & Neuro-inflammation
Modulating the HPA axis and sympathetic nervous system to lower pro-inflammatory cytokines.
Improve Brain Plasticity
Strengthening connectivity within and between neural networks impacted by PD.
Address Motor Control
Dedicated interventions focused on gait, movement, speech therapy, and fine motor skills.
Rebalance Emotion & Cognition
Psychological interventions to reduce stress, anxiety, and depression while building resilience.
Gamification & Reward Systems
Techniques to maintain reward system health and reinforce positive neuro-immunological responses.



Clinically Validated
& Mechanistically Proven
We measure hard biological endpoints to prove the hybrid effect. Our Phase 2 results demonstrated significant improvements in the Unified Parkinson's Disease Rating Scale (UPDRS) Parts 2 & 3 compared to standard of care. Furthermore, fMRI imaging confirmed our ability to activate specific neural circuits through digital intervention.
fMRI Imaging
Visualizing changes in brain connectivity and structure.
Biomarkers
Analyzing blood and saliva for markers of inflammation (Cortisol, Cytokines).
Standardized
Clinical ScalesUsing the gold-standard metrics for each disease state (e.g., MDS-UPDRS for Parkinson's).
Join our. Clinical Trials.
Remepy is currently conducting clinical trials for Parkinson’s Disease patients. If you are interested in learning more about our clinical trials, contact us today.







